How was the electron discovered?
J.J Thomson

http://www.nobelprize.org/nobel_prizes/physics/laureates/1906/thomson.jpg
One of the greatest thinkers and physicists of the late 19th and early 20th centuries, Sir J. J. Thomson is a British native.
Exclusive office interview with Thomson!
Alice: Good morning, Mr. Thomson. It's such a pleasure to meet you!
Thomson: Why thank you, it's a pleasure to meet you too. I'm happy to answer any questions you might have about my work.
A: Your recent discoveries about the depths of the atom have made big headlines. Would you mind telling us what they are exactly?
T: Well, through my experimenting, I discovered the electron and developed a new model of the atom. I've managed to shed some light about how the universe works.
A: Great! Why don't you tell us more about the electrons?
T: Sure. Back in 1897, I was experimenting with cathode rays at Cambridge University. Cathode ray tubes are simple glass tubes with wires at opposite ends. When we pump out the air and put a high voltage across it, a curious fluorescent light emits from it. Many of my contemporaries have tried to explain this mysterious glow. It made sense that these rays were similar to normal light waves, which they thought traveled in "ether", an invisible fluid. It also made sense to me that the rays were made up of material particles, which I thought were all made up of ether anyway. My contemporaries had already experimented with their ideas.
Thomson: Why thank you, it's a pleasure to meet you too. I'm happy to answer any questions you might have about my work.
A: Your recent discoveries about the depths of the atom have made big headlines. Would you mind telling us what they are exactly?
T: Well, through my experimenting, I discovered the electron and developed a new model of the atom. I've managed to shed some light about how the universe works.
A: Great! Why don't you tell us more about the electrons?
T: Sure. Back in 1897, I was experimenting with cathode rays at Cambridge University. Cathode ray tubes are simple glass tubes with wires at opposite ends. When we pump out the air and put a high voltage across it, a curious fluorescent light emits from it. Many of my contemporaries have tried to explain this mysterious glow. It made sense that these rays were similar to normal light waves, which they thought traveled in "ether", an invisible fluid. It also made sense to me that the rays were made up of material particles, which I thought were all made up of ether anyway. My contemporaries had already experimented with their ideas.
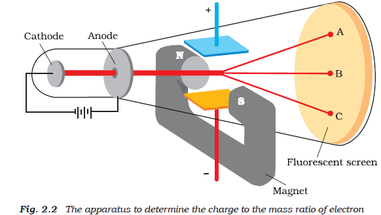
T:An easy experiment that I can show you right now is putting a magnet close to the cathode ray tube. The magnet is actually able to bend the rays and push it around the tube.
A: Wow! That seems like evidence that the rays are made up of particles, because for the rays to be pushed around, they have to be made up of a matter, right? T: Yes, but it wasn't enough to prove it. I had to experiment further. In 1895, Jean Perrin concluded that these rays had a negative charge.I wanted to see if it was possible to separate the ray from its charge, so I set up my first experiment. I connected the end of the tube with metal cylinders and an electrometer, a device that is able to measure electrical charge. |
T: When I bent the ray with a magnet so that the ray entered the the cylinders and electrometer, it measured a significant amount of electric charge. When I pushed the ray so that it didn't enter the cylinder, it measured very little charge.
A: So you couldn't separate the ray from its charge? Interesting. So is the ray a charged particle?
T: Well, I concluded that the charge and the rays must be bound together somehow, and it did lead me ask to that question. Since charged particles bend in the presence of an electric field, my next experiment was to find out if I could bend the rays with one. Many scientists had already tried this experiment, but I thought that the remnants of the gas inside the tube was acting as a conductor, which counteracts the effect of the electrical field. So, I removed as much of the gas as was possible, and voila! The electric field actually bent the ray!
A: So you couldn't separate the ray from its charge? Interesting. So is the ray a charged particle?
T: Well, I concluded that the charge and the rays must be bound together somehow, and it did lead me ask to that question. Since charged particles bend in the presence of an electric field, my next experiment was to find out if I could bend the rays with one. Many scientists had already tried this experiment, but I thought that the remnants of the gas inside the tube was acting as a conductor, which counteracts the effect of the electrical field. So, I removed as much of the gas as was possible, and voila! The electric field actually bent the ray!
A: Incredible! So are the ray and its charge the same thing?
T: I didn't know. My conclusion was that cathode cathode rays are negative charges carried by material particles. I couldn't know for sure what these particles (I called them corpuscles) actually were until I experimented again. In my last experiment, I found out the ratio of the mass of a particle to its charge. To do this, I bent the ray with a magnet and measured how much they were bent, and also measured how much energy the ray carried. I calculated the charge-to-mass ratio of one of these particles to be 1.758820150(44)×10^11 C/kg, which means that there is a huge negative charge on a extremely small particle. I was surprised at this - it meant that these particles must be extremely small. "Could anything at first sight seem more impractical than a body which is so small that its mass is an insignificant fraction of the mass of an atom of hydrogen?" I was convinced that these particles were the basis of matter, including atoms.
T: I didn't know. My conclusion was that cathode cathode rays are negative charges carried by material particles. I couldn't know for sure what these particles (I called them corpuscles) actually were until I experimented again. In my last experiment, I found out the ratio of the mass of a particle to its charge. To do this, I bent the ray with a magnet and measured how much they were bent, and also measured how much energy the ray carried. I calculated the charge-to-mass ratio of one of these particles to be 1.758820150(44)×10^11 C/kg, which means that there is a huge negative charge on a extremely small particle. I was surprised at this - it meant that these particles must be extremely small. "Could anything at first sight seem more impractical than a body which is so small that its mass is an insignificant fraction of the mass of an atom of hydrogen?" I was convinced that these particles were the basis of matter, including atoms.
Hear Thomson Speak for himself in this recording:
Thomson's Plum Pudding Model

Picture source: http://www.kentchemistry.com/links/AtomicStructure/plum.gif
A: Interesting! So that's how you discovered the electron! However, didn't your findings clash with Dalton's atomic model? How can atoms be made up of these particles?
T: Well, that's where my plum pudding model comes in. I developed it in 1904 to show that there are negatively charged particles within the atom. To counteract this negativity, the rest of the atom must be positively charged. It makes more sense to have these corpuscles floating like plums in a pudding. And yes, my findings disprove the notion that atoms are indivisible, rather, they are made up of even smaller parts.
A: Well, that's all I have time for, thanks for explaining everything!
T: Well, that's where my plum pudding model comes in. I developed it in 1904 to show that there are negatively charged particles within the atom. To counteract this negativity, the rest of the atom must be positively charged. It makes more sense to have these corpuscles floating like plums in a pudding. And yes, my findings disprove the notion that atoms are indivisible, rather, they are made up of even smaller parts.
A: Well, that's all I have time for, thanks for explaining everything!